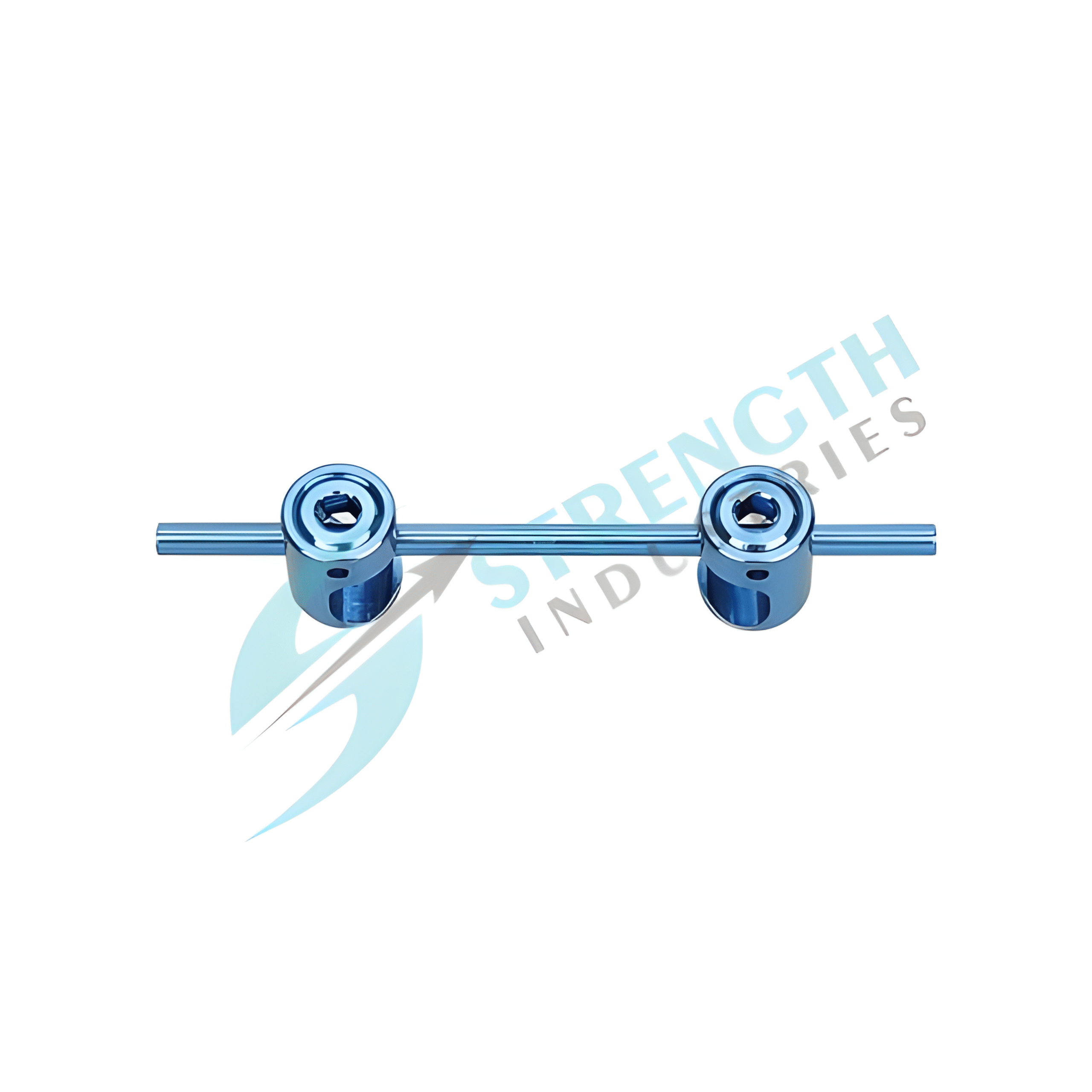
The 6.0 Crosslink-II is an advanced spinal implant accessory, designed to provide enhanced torsional stability and rigidity in spinal fixation procedures. It ensures secure interconnection between rods, reducing micro-motion and improving the overall effectiveness of spinal stabilization.
Technical Features & Specifications:
- Material: Premium Surgical Stainless Steel / Titanium Alloy
- Rod Compatibility: 6.0mm
- Available Sizes: 60mm, 70mm, 80mm
- System Design: Strong, lightweight, and corrosion-resistant
- Certifications: ISO 13485 & CE approved
Clinical Applications & Uses:
- Provides cross-stability in spinal fixation systems
- Minimizes rotational instability and micro-motion
- Supports deformity correction and trauma reconstruction
- Used in degenerative and scoliosis corrective surgeries
Advantages & Benefits:
- Improved Stability: Rigid interconnection between rods
- Durable Construction: Made with surgical-grade materials
- Optimized Design: Lightweight yet strong for ease of use
- Multiple Options: Available in 60mm, 70mm, and 80mm lengths
- ISO & CE Certified: Globally approved for surgical use
Available Configurations & Sizes:
| Code | Size (mm) | Rod Diameter | System Type |
|---|---|---|---|
| 2100-2101 | 60mm | 6.0mm | Crosslink-II |
| 2100-2102 | 70mm | 6.0mm | Crosslink-II |
| 2100-2103 | 80mm | 6.0mm | Crosslink-II |
Why Choose Us?
- Precision Engineering: Manufactured with advanced CNC technology
- Global Standards: ISO 13485 & CE certified
- Surgeon Trusted: Reliable implant used by orthopedic experts
- Superior Materials: High biocompatibility and corrosion resistance

















Reviews
There are no reviews yet.